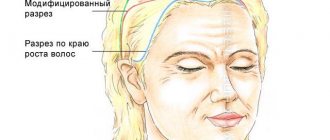
Deciding to have breast augmentation surgery is not as easy as it might seem. One of the reasons is women's frequent concerns about the quality of breast implants. On the Internet and from friends, you have probably heard stories and admonitions about the dangers of implant rupture and the unpleasant consequences of such an event. In fact, these cases are very rare, but you should still pay close attention to the quality of the silicone prosthesis, the reliability of the manufacturer and product reviews from real patients.
Today, many companies offer breast implants, but it can be difficult to choose, because the products are not on everyone’s lips. We suggest paying attention to Allergan implants, which are of the highest quality.
Let's name just three facts that can convince you of the reliability of the product:
- Allergan is the inventor of the drug Botox, successfully used throughout the world,
- The manufacturer provides a lifetime warranty on Allergan breast implants,
- Products are supplied only to certified medical institutions.
What's special about Allergan implants?
Of course, every woman wants her breasts to look beautiful. Allergan takes the development of breast implants very seriously - they have various shapes suitable for a particular breast type, have several degrees of shell protection, and have an anti-slip surface that prevents the prosthesis from moving inside the body. You can see the main shapes and sizes in the table below; each brand has dozens or hundreds of specific models.
Table of Natrel Allergan implants
According to reviews of women who have undergone breast surgery with Natrel Allergan implants (a lot of comments can be found on the Internet, including photos), the breasts both look and feel as close to natural as possible.
Absence of unnatural bumps, excessive skin tension, hardness of the breast - these are the main problems of improper prosthetics and the choice of cheap products for plastic surgery. "Allergan", on the contrary, has not been noticed in such undesirable consequences of the operation. Of course, the professionalism of the surgeon should also be taken into account.
The case of Allergan Natrelle implants: they are recalled due to the risk of breast cancer. What to do?
What happened: Allergan, a market leader in plastic surgery and cosmetics, recalled Natrelle breast implants. They did this after the US Food and Drug Administration (FDA) analyzed 660 reports of cancer in patients who received breast implants from 2010 to September 2021 and found 457 cases of anaplastic large cell lymphoma, of which 9 - with fatal outcome.
To the question of how this became possible and what should those who have these implants do now, answers Alexander Andreev, a plastic surgeon at the Institute of Plastic Surgery and Cosmetology.
Alexander Andreev
Plastic surgeon at the Institute of Plastic Surgery and Cosmetology
Allergan Natrelle breast implants are filled with a cohesive gel and have a macrotextured (rough) shell. This surface allows the tissues to grow into the implant shell and be securely fixed there. According to new research, it is this membrane that provokes the development of a very rare type of cancer - anablastic large cell lymphoma (ALCL).
In my opinion, the situation is very ambiguous. The probability of developing the disease, according to various estimates, is very low - about 1:25,000. In Russia, two such cases were recorded over ten years of observation. During this time, several tens of thousands of similar implants were installed.
All implants used in our country as medical devices undergo strict certification by inspection bodies to confirm their safety for patients.
Allergan implants have also been FDA certified in the USA and have also been found safe for use in Russia and Europe
They were among the most popular in the world. Evidence that implants can cause ALCL has only recently begun to emerge, although the company has been on the market for decades. When enough information had accumulated, the company decided to recall the products. But again, the likelihood of complications developing is extremely low.
Recommendations for those who already have these implants installed are the most banal: regularly do ultrasound of the mammary glands and visit a mammologist at least once a year.
This is quite enough to diagnose all possible complications. Moreover, this applies to patients who have implants installed not only from Allergan, but also from any others. It is important to get checked even for those who have not had surgery at all. Take care of your health, because any disease is much easier to cure in its initial stage.
Significant changes in the cost of operations should not be expected due to the scandal. There are a lot of implant manufacturers, and their prices are approximately the same.
Allergan is also prescribed for medical reasons
Not only the desire to restore youth, but to correct the shortcomings of nature drives women to the decision to undergo plastic surgery on the breast. Many congenital or medical problems require restoration or correction of the female body in its most attractive part. So: most often, Allergan brand products are used to restore aesthetics or correct for such indications. There are several reasons for this:
- anatomical shape,
- the ability to make almost any breast shape,
- combining different types of gel inside the breast implant to achieve naturalness,
- different profile heights, suitable for different types of breasts and depending on their condition.
In our clinic you can get more detailed professional advice on the use of products from this brand for breast surgery. Sign up for a consultation by phone and make your choice. Call right now and make an appointment at a time convenient for you.
Next article Previous article
Allergan implant recall: reasons and algorithm for patient actions
The global community of plastic surgeons is discussing the latest news regarding mammoplasty. Allergan announced a worldwide recall of textured breast implants Wednesday after the U.S. Food and Drug Administration (FDA) found a sharp increase in rare cancer cases and deaths associated with the products and asked the company to remove them. from the US market.
The Dublin-based company said it is recalling Biocell textured breast implants and tissue expanders from all markets in which they are sold. Endoprostheses have previously been banned or recalled in Europe and Canada.
The FDA says new global data has linked 573 cases of a rare cancer to implants since the agency began tracking the problem back in 2011. The vast majority of these cases involve Allergan products. 33 women died from what is known as breast implant-related anaplastic large cell lymphoma (ALCL). Of those deaths, authorities identified the manufacturer of the implants in 13 cases—and it was Allergan in all but one.
The US agency will continue to evaluate any new information and may take action regarding other breast implants as a result if it is warranted. Although most cases of anaplastic lymphoma are associated with the use of textured breast implants, particularly macrotextured implants such as Allergan implants, the collaborators will continue to study to determine whether the risk of developing ALCL is limited to specific models of textured breast implants or all breast implants.
Following the agency's release of the data, Allergan said it was initiating a voluntary recall worldwide "as a precautionary measure following notification of recently updated global safety information regarding the unusual incidence of [ALCL] provided by the FDA."
The company has asked healthcare professionals to immediately stop using these implants in clinical practice and return any unused products. The recall affects only Biocell products from Allergan. Natrelle and Microcell smooth implants and tissue expanders will remain on the market.
Allergan has provided a detailed list of products subject to removal from the global market in the following table:
What is anaplastic large cell lymphoma?
ALCL is not breast cancer, it is a type of non-Hodgkin's lymphoma (cancer of the immune system). In most cases, palologia is found in scar tissue and fluid near the implant, but in some cases it can spread throughout the body. Currently, the overall incidence of ALCL is considered extremely low; however, this diagnosis is serious and can lead to death, especially the time for timely diagnosis and treatment has been lost. In most patients, this type of lymphoma is successfully treated surgically by removing the implant and scar tissue surrounding the implant; however, some patients may require treatment with chemotherapy and/or radiation therapy.
What should patients who have Allergan Biocell implants do?
If you don't have any symptoms, experts do not recommend removing these or other types of breast implants due to the low risk of developing anaplastic large cell lymphoma. However, if you have any questions, talk to your healthcare provider.
Know the symptoms of ALCL. The first is persistent swelling or pain near the breast implant. Monitor the area around your breast implants for any changes.
If you experience any of these symptoms or notice other changes, talk to your healthcare provider about needing an evaluation. Typically this refers to a physical examination, imaging and/or evaluation of the fluid or tissue around the breast implant.
If you don't have any symptoms, experts do not recommend removing these or other types of breast implants
Based on consultation with your healthcare provider, patients with confirmed ALCL should undergo surgery to remove the breast implant and excision of the surrounding scar capsule, which is a more extensive operation than isolated removal of the implant alone.
As with any implanted device, it is always a good idea to keep a record of the device, recording information about the unique identifier and model name of the implant. You may have been given this information along with your implants. If you want any of this information, try asking your surgeon or getting a record of your surgery from the facility where it was performed.
It should be understood that most cases of ALCL occur many years after breast implant placement. Talk to your surgeon about your risk of developing the disease.
Features of MENTOR® silicone implants (“Mentor”)
- Endoprostheses for breast correction are manufactured using Memory Gel, a patented filler gel. This silicone has shape memory, that is, the implant instantly restores its previous shape after any mechanical impact.
- The safety of these breast implants has been proven in numerous clinical studies, and the FDA approved MENTOR® products back in 1992. The company provides a lifetime warranty on its products.
- Memory Gel has a special viscosity, which, on the one hand, allows you to recreate the natural density of the mammary glands, and on the other, does not leak if the membrane is damaged.
To reduce the incidence of negative consequences of breast correction operations associated with the development of postoperative contracture, Mentor produces implants with both smooth and textured surfaces.
The textured surface of Mentor implants is worth talking about separately. The endoprosthesis shell is made using Siltex® technology, using the inprinting method, and has a moderately textured surface, which ensures optimal engraftment of the implant with the surrounding tissues after installation.
Thanks to this feature, the risk of capsular contracture is reduced: a natural fibrous membrane is formed around the implant, just like around any foreign body; if it becomes too dense, then this is reflected both in appearance (the implant is palpable and becomes noticeable, the breast is deformed) and on the patient’s health.
1. ROUND IMPLANTS – Implants of this shape allow you to achieve a light and seductive push-up effect, as well as filling the upper pole of the mammary glands.
2. ANATOMICAL IMPLANTS – They are also called “teardrop” because they follow the smooth slope of the mammary glands with an unfilled upper pole. It is believed that compared to round implants, this shape looks more natural.
Breast implants may cause cancer
1K 2 min.
Breast implant manufacturer Allergan announced that it is stopping supply and recalling already supplied breast implants that have a textured surface.
Photo: Daniel Reinhardt / DPA / TASS
Photo: Daniel Reinhardt / DPA / TASS
This decision was dictated by data from the American Food and Drug Administration (FDA): it is implants manufactured by Allergan (trademark Biocell) with a relief surface that cause the majority of cases of a rare form of lymphoma. The same information was received from the supervisory authorities of France, Australia and Canada.
However, the FDA has not issued a recommendation for women with existing implants to remove them: the cancer is very rare, but they should consult a doctor if they experience any unpleasant symptoms, such as pain or swelling.
The relief surface was invented for Biocell implants to prevent freedom of their movement in the mammary gland after implantation, in addition, the relief surface minimizes the appearance of scar tissue. The brand occupies only 5% of the US market. The vast majority of breast implants used in the United States have a smooth surface.
The Biocell recall does not in any way affect the supply of smooth implants manufactured by Allergan, nor textured implants under another brand, Microcell.
For the first time, regulatory authorities linked Biocell implantation with the occurrence of lymphoma back in 2011. The paradox is that the reason for the appearance of implants in women is often reconstructive surgery after partial removal of the mammary gland due to a malignant neoplasm. Lymphoma is not breast cancer; although the disease is malignant, it develops slowly and is usually successfully cured by surgery, including removal of implants.
In May of this year, the FDA was more supportive and did not require Allergan to recall products and stop supplying them. But now the direct connection of this particular implant model with lymphoma has become obvious, and Biocell had to be banned.
The connection between other embossed implants and cancer has not been established.
“Once a particular manufacturer's product was shown to be highly likely to be directly associated with significant patient harm, including death, the FDA took action,” explained FDA Deputy Administrator Amy Abernethy.
More than 80% of the 570 confirmed cases of this type of lymphoma worldwide have been linked to Allergan implants, according to the FDA. Regulators estimate that the risk of disease is six times higher with Allergan implants than with contoured implants from other manufacturers.
FDA officials said they decided to implement the ban after they received 116 new reports of lymphoma and the number of deaths from the disease reached 33, including at least 12 cases in women with Allergan breast implants.
Anatoly Krivov